 
Solutions of isosorbide dinitrate (200 μg/mL) and diazepam (7.5 μg/mL) were prepared by dissolving in an isotonic saline solution (0.9%NaCl, 500mL Viaflo ®, Baxter, Maurepas, France) in a glass vial before using immediately. I0775000, vial of 350 mg, Council of Europe-EDQM, Strasbourg, France) and diazepam (Diazepam, vial of 25 g, Cooper, Melun, France). Impact of infusion device on drug deliveryĭrug solutions were prepared from powder forms of active pharmaceutical ingredients: isosorbide dinitrate (Isosorbide Dinitrate CRS, European Pharmacopoeia Reference Standard, ref. Īs many multi-lumen extension tubes with different materials and sterilisation processes are available, the purposes of our study were, firstly, to measure the concentration of two drugs after passing through devices available on the market and, secondly, to determine the nature of the PUR used in the infusion devices and its impact on drug delivery. Studies have shown that PUR provides good irradiation resistance thanks to its high degree of crosslinking. The susceptibility of plastics to the process of high energy radiosterilisation has been shown to be closely dependent on the chemical structure and purity of the material. Sterilisation techniques can either act physically or chemically leading to alterations in the structure of macromolecules, which can result in chain scission, oxidation, crosslinking, melting or hydrolysis. Other criteria have to be taken into account, especially the mechanical properties of the materials, their ease of assembly and their manufacturing costs.ĭifferent techniques are used to sterilise infusion devices (ethylene oxide (EtO) gas or gamma irradiation (GI)) and these may have harmful effects on medical grade polymers such as extensive material degradation and plastic deformation/modification affecting drug-device interaction. 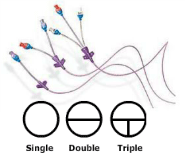
Plasticised PVC has high potential interaction with drugs, whereas PE or PP materials are subject to less. The nature of the plastic material is an important parameter because it determines the type and the amount of drug binding. This phenomenon leads indeed to drug loss and results in reduced drug delivery to the patient. The evolution in infusion device materials may affect drug-device interaction and so drug delivery, especially with possible drug adsorption on the inner plastic surface of the container. DEHP has been replaced by alternatives and in some cases, plasticised PVC has been replaced by other materials. However, its composition has changed recently with doubts concerning the use of di-(2-ethylhexyl) phthalate (DEHP) and other hazardous phthalate plasticisers. Plasticised PVC is the most common material for infusion devices. Plastic materials such as polyurethane (PUR), polyethylene (PE), polypropylene (PP) and polyvinyl chloride (PVC) are widely used in various medical devices. However, other characteristics of the device affect drug delivery and have to be taken into account. This parameter is related to the delay to reach the steady state in drug delivery after a change in drug flow during multi-infusion therapy. The main criterion of choice for these devices appears to be their dead volume, defined as the volume between the meeting point of the drugs simultaneously infused and the egress of the device. Patients in intensive care units receive many drugs simultaneously, resulting in the need for multi-lumen extension tubes. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.ĭata Availability: All relevant data are within the paper.įunding: This study was funded by intramural funds from the Department of Biopharmacy, Galenic and Hospital Pharmacy, Lille 2 University, UDSL, EA GRIIOT, UFR Pharmacie, Lille, France.Ĭompeting interests: Aurélie Foinard, Bertrand Décaudin, and Pascal Odou report receiving travel-expense reimbursements from Doran International. Received: JanuAccepted: ApPublished: May 6, 2016Ĭopyright: © 2016 Maiguy-Foinard et al. PLoS ONE 11(5):Įditor: Abdelwahab Omri, Laurentian, CANADA Citation: Maiguy-Foinard A, Blanchemain N, Barthélémy C, Décaudin B, Odou P (2016) Influence of a Double-Lumen Extension Tube on Drug Delivery: Examples of Isosorbide Dinitrate and Diazepam.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
